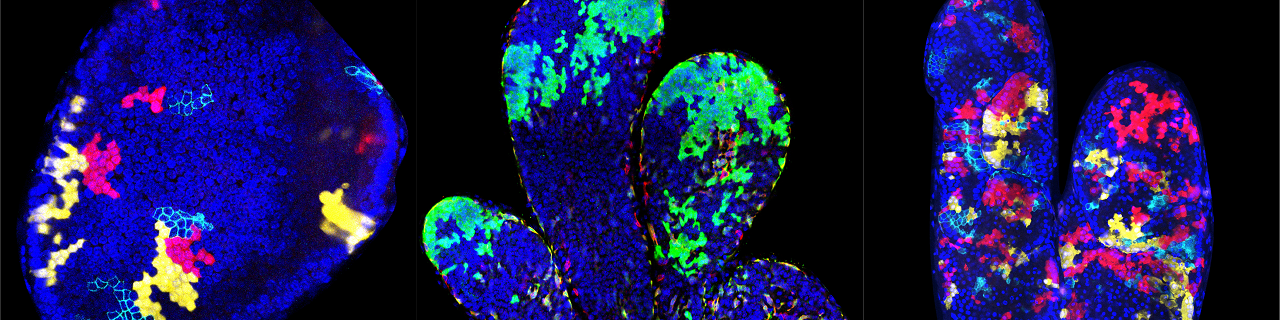
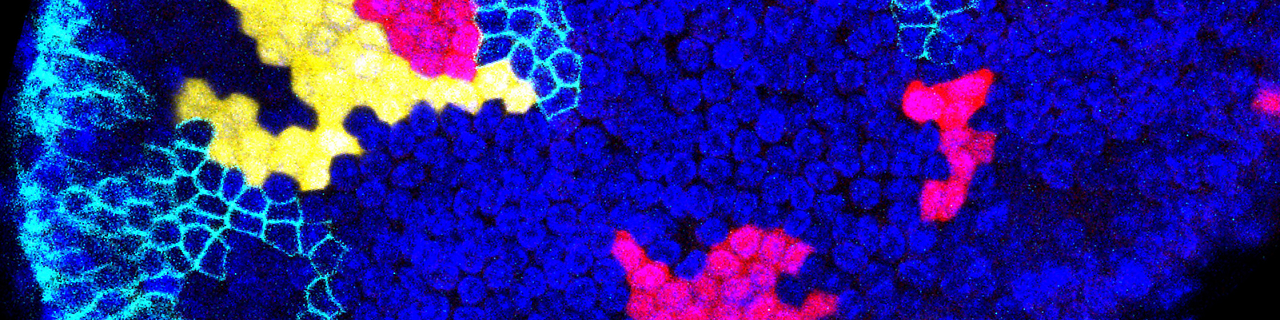
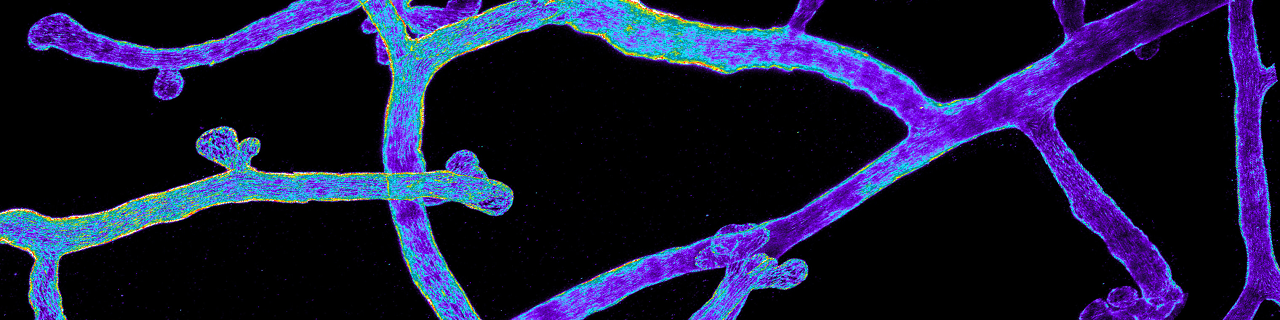
Stem cells of the mammary gland and the prostate
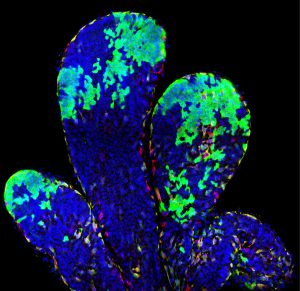
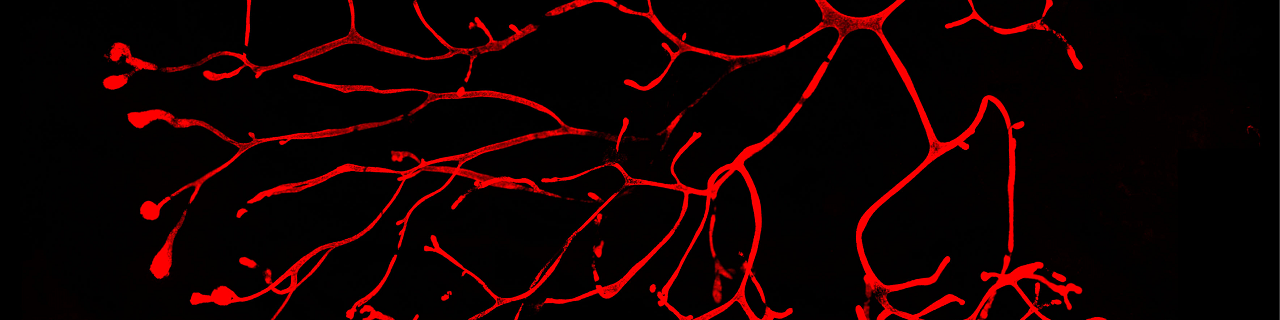
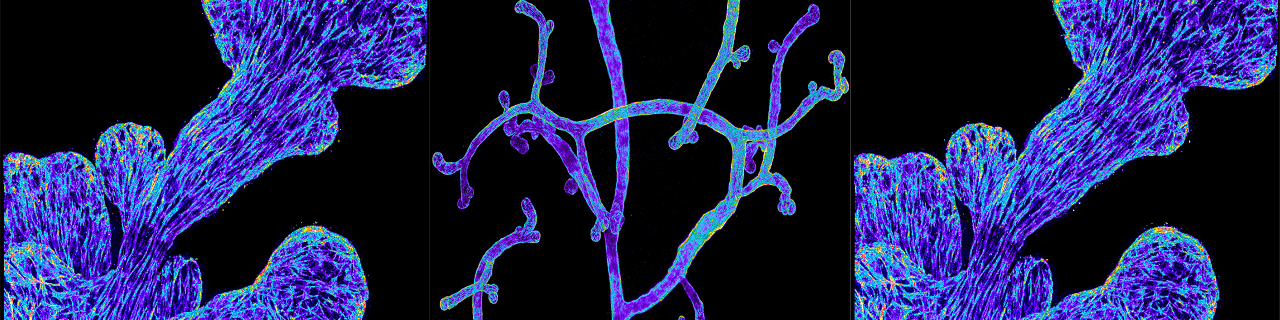
The mammary glands are essential during early postnatal life by providing to young mammalian offspring nutrients, water and electrolytes, and immune defense until they can survive independently. The mammary glands are epidermal appendages that progressively sprout and branch to invade the underlying mesenchyme. At puberty, the mammary gland expands considerably to form a highly branched tubular structure. Each tube is composed by one inner layer of luminal cells facing the lumen, surrounded by an outer layer of myoepithelial/basal cells in contact with the basal lamina. During pregnancy and lactation, the mammary glands expand again and undergo profound remodeling, ensuring the development of the alveolar structures, which are responsible for the production of milk. The ability to replenish the mammary gland through cycles of pregnancy had been attributed to the existence of mammary gland stem cells.
Our lab is interested in understanding the mechanisms regulating the fate of mammary gland stem cells.
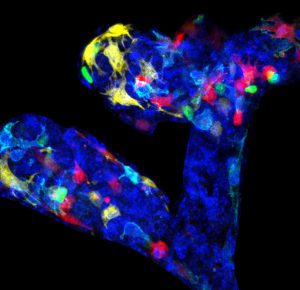
Using novel lineage tracing of basal and luminal mammary gland cells during embryonic and postnatal development as well as during pregnancy and lactation, we demonstrated that the mammary glands initially develop from embryonic multipotent stem cells that are replaced soon after birth by unipotent basal and luminal ER+ and ER- lineage restricted stem cells that mediate postanatal development and adult homeostasis (Nature 2011, Genes and Development 2016, Cell Reports 2017, Nature Cell Biology 2018).
We demonstrated that unipotent basal stem cells identified by lineage tracing become multipotent and able to reform the different mammary gland lineages following their transplantation into the mammary mesenchyme, demonstrating the important plasticity of the mammary gland stem cells during regenerative conditions (Nature 2011).
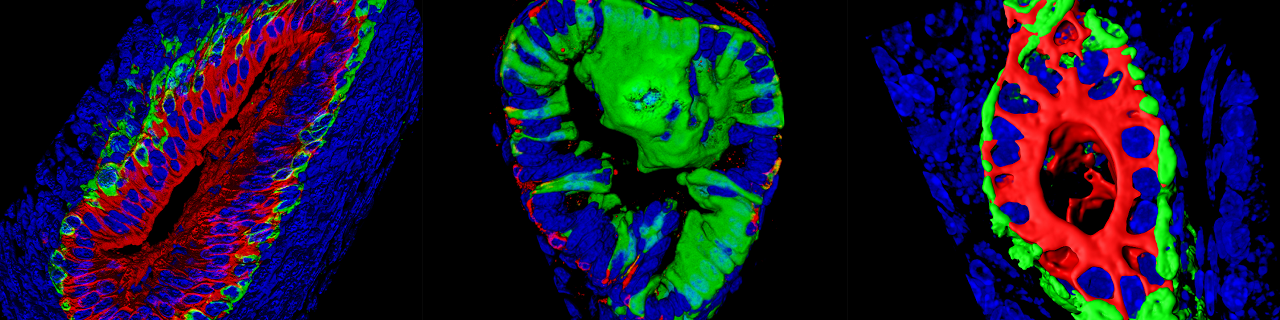
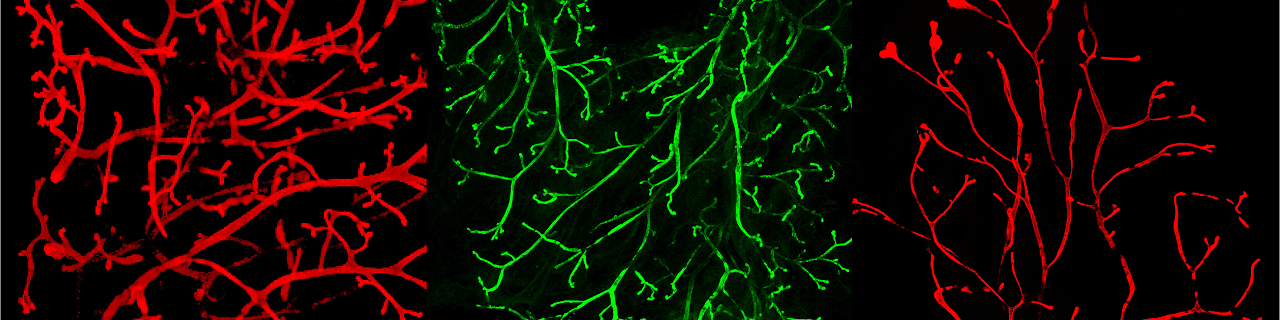
Using similar lineage tracing approaches, we demonstrated that prostate postnatal development is mediated by multipotent basal cells that becomes unipotent in adult life (Nature Cell Biology 2012, Genes and Development 2016).
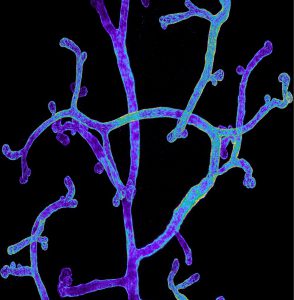
We demonstrated that active signaling arising from luminal cells restricts basal cell multipotency (Centonze Nature 2020).
People
Alexandra Van Keymeulen, permanent FNRS researcher
Chen Jiang, postdoctoral researcher
Yura Song, PhD student
Brigida Novello, PhD student
Viaene Amélie, PhD student
Gaëlle Bouvencourt, technician
Zahra Zahedi, technician
Selected publications
Distinct stem cells contribute to mammary gland development and maintenance. Van Keymeulen A, Rocha AS, Ousset M , Beck B, Bouvencourt G, Rock J , Sharma N, Dekoninck S & Blanpain C. Nature 2011 Oct 19;478(7369):399-403. Highlighted by previews in Nature Cell Biology, Cell Stem Cell, and EMBO J.
Multipotent and unipotent progenitors contribute to prostate postnatal development. Ousset M, Van Keymeulen A, Bouvencourt G, Sharma N, Achouri Y, Simons BD, Blanpain C. Nat Cell Biol. 2012 Nov;14(11):1131-8. Highlighted by a preview in EMBO Reports
Plasticity of epithelial stem cells in tissue regeneration Blanpain C & Fuchs E. Science. 2014 Jun 13;344(6189):1242281.
Reactivation of multipotency by oncogenic PIK3CA induces breast tumor heterogeneity. Van Keymeulen A, Lee MY, Ousset M, Rorive S, Brohée S, Wuidart A, Bouvencourt G, Giraddi R, Dubois C, Salmon I, Sotiriou C, Phillips WA, Blanpain C. Nature 2015 Sep 3;525(7567):119-23. Highlighted by a preview in Cancer Discovery
Quantitative lineage tracing strategies to resolve multipotency in tissue specific stem cells. Wuidart A, Ousset M, Rulands S, D Simons BD, Van Keymeulen A, Blanpain C. Genes Dev. 2016 Jun 1;30(11):1261-77. Cover article
Lineage-Restricted Mammary Stem Cells Sustain the Development, Homeostasis, and Regeneration of the Estrogen Receptor Positive Lineage. an Keymeulen A, Fioramonti M, Centonze A, Bouvencourt G, Achouri Y, Blanpain C. Cell Rep. 2017 Aug 15;20(7):1525-1532.
Early lineage segregation of multipotent embryonic mammary gland progenitors. Wuidart A, Sifrim A, Matsumura S, Fioramonti M, Brisebarre A, Brown D , Centonze A, Dannau A, Dubois C, Van Keymeulen A, Voet T, Blanpain C. Nat Cell Biol. 2018 Jun;20(6):666-676. Cover article Highlighted by a news and views in Nature Cell Biology
Spatiotemporal regulation of multipotency during prostate development. Tika E, Ousset M., Dannau A, Blanpain C. Development 2019 Oct 23;146(20). doi: 10.1242/dev.180224.
Heterotypic cell-cell communication regulates glandular stem cell multipotency. Centonze A, Lin S, Tika E, Sifrim A, Fioramonti M, Malfait M, Song Y, Wuidart A, Van Herck J, Dannau A, Bouvencourt G, Dubois C, Dedoncker N, Sahay A, de Maertelaer V, Siebel CW, Van Keymeulen A, Voet T, Blanpain C. Nature. 2020 Aug;584(7822):608-613. Epub 2020 Aug 26. Cover article.
Collagen signaling and matrix stiffness regulate multipotency in glandular epithelial stem cells in mice. Jiang C, Centonze A, Song Y, Chrisnandy A, Tika E, Rezakhani S, Zahedi Z, Bouvencourt G, Dubois C, Van Keymeulen A, Lütolf M, Sifrim A, Blanpain C. Nat Commun. 2024 Dec 18;15(1):10482. doi: 10.1038/s41467-024-54843-5.